Deepwater Program: Exploration and Research of Northern Gulf of Mexico Deepwater Natural and Artificial Hard Bottom Habitats with Emphasis on Coral Communities: Reefs, Rigs and Wrecks
Background
Introduction
 This program is a $3.7M four-year project from the Bureau of Ocean Energy Management (BOEM) in August 2008. This is an interagency study involving BOEM and the National Oceanic and Atmospheric Administration (NOAA) Office of Ocean Exploration and Research (OER) and will also include additional collaboration with the U.S. Geological Survey.
This program is a $3.7M four-year project from the Bureau of Ocean Energy Management (BOEM) in August 2008. This is an interagency study involving BOEM and the National Oceanic and Atmospheric Administration (NOAA) Office of Ocean Exploration and Research (OER) and will also include additional collaboration with the U.S. Geological Survey.
This is a similar type project to the BOEM Chemo-III project that is currently underway at TDI-BI. It will be multidisciplinary with principal investigators from TAMU, Penn State, LSU, TAMUCC and WHOI.
Up to twenty-four (24) days of ship time the first year and similar number of days for years two and three aboard NOAA OER-funded research vessels including operating personnel, made available by the NOAA OER, will be used to conduct this program. Other research vessels, remotely operated vehicles (ROV), human occupied vehicles (HOV) and autonomous underwater vehicles (AUV) from NOAA OER may also be employed in FY 2009 and 2010. The first cruise is scheduled for September 2008.
This study will focus on the exploration and investigation of deepwater non-chemosynthetic biological communities in the deep (>300 m) Gulf of Mexico (GOM) that have developed on both natural and artificial hard substrates with emphasis on deepwater coral habitats. An additional aspect includes exploration of shipwreck sites in the deep Gulf with historical and biological objectives.
Scope and Objectives
- Discover and describe new locations at greater than 300m depth in the GoM with extensive coral community development, particularly including Lophelia pertusa.
- Gain a more comprehensive understanding of the fundamental processes that control the occurrence and distribution of Lophelia and other extensive coral communities at depths greater than 300 m in the GoM through both laboratory experiments and field data collection.
- Document and understand the relations between coral communities on artificial and natural substrates with respect to community composition and function, phylogeographic and population genetics, and growth rates of the key cnidarian foundation fauna.
Our specific biological objectives:
- To discover and characterize new sites:
- Characterize key sites at the largest scale with high resolution bathymetry, side scan sonar imaging, 3D seismic data, and current models
- Characterize the coral density at all study sites at the 10 to 100 m scale with randomized photo transects and general site descriptions.
- Characterize the community composition at the 1 to 10 m scale at significant coral sites (man-made and natural) with analysis of close up imagery, replicate 5x5m photomosaics, and quantitative community collections.
- Analyze connectivity among all sites in the GoM (man-made and natural), and to other sampled populations, with comparative community, phylogeographic and population genetic analyses.
- Compare the structure, species richness and diversity of communities tightly associated with Lophelia on man-made structures and from natural sites in the GoM
- Experimentally determine the tolerance and growth response of Lophelia to temperature, pH/alkalinity, dissolved oxygen and electrical current.
- Characterize and constrain growth rates of key species of colonial cnidarians (pioneer colonies) using analyses of images of the largest colonies on man-made structures of known age.
- Characterize key variables at sites with the most significant communities of coral colony development over one year by monitoring temperature, currents, larval seasonal distribution and sediment quality and quantity at 2-4 sites between 2009 and 2010.
By integrating this information, we will develop a predictive model to examine the potential occurrence of significant assemblages of L. pertusa or other cnidarians at unexplored sites and artificial reefs in the deep GoM.
Historic Shipwreck Component
The proposed study will be multidisciplinary in scope. It will focus on the archaeological and biological aspects of up to six shipwrecks in the north-central portion of the GoM. Water depths at the designated investigation sites range from 554m to 2,286m. Each shipwreck will be investigated to determine identity, site boundaries, National Register eligibility, preservation state and stability, associated biological communities, and an artificial reef effect on meiofauna and/or mobile fish communities.
Our specific archaeological objectives:
1) To record each vessel through detailed imagery to establish its type, date of construction, and positive identification if possible.
2) To establish nationality, ownership (past and present), use history, cause of loss, mission and cargo at time of loss through fieldwork and historical research.
3) To determine the extent and condition of the artifact assemblage on each vessel and the presence of diagnostic artifacts.
4) To determine potential eligibility to the National Register of Historic Places through archival research and the analysis of imagery and to prepare a National Register nomination form for potentially eligible vessels.
5) To assess impacts of biofouling communities to these shipwrecks to determine the stability of these sites and rate of deterioration.
Our specific bacteriological objectives:
1) Determine the rates of deterioration of test coupons placed on platforms and sited on specific shipwrecks that have already been visited and were installed on previous expeditions to those sites of interest. Determination may take the form of passive observations of the coupon condition within the platform of interest. If significant deterioration rates have already occurred then the platform may be recovered for on-ship and in-laboratory investigations.
2) Fresh test platforms may be deployed to either replace recovered platforms or to determination the deterioration rate at a fresh site of archeological significance.
3) Examine recovered natural samples from deteriorating sites and localized waters that may contain active bacterial communities traditionally associated with corrosive or bio-concretious growths.
4) On selected recovered test coupons conduct such experiments as would allow a more precise projection of the rates of deterioration of test material through the rates of pitting (in the event of mild steel) to losses in physical composition (in the event of cloths and wood.
5) Recovered bacteriological communities from the sites of interest will be subjected to laboratory analysis to determine major bacterial communities, degrees of activity (using the ATP methodology), and identification of community structures by fatty acid methyl ester analysis (MIDI/BART technologies).
Directed Missions of BOEM and NOAA OER
Increased oil and gas activity beyond the shelf break and US recognition of the 200 m EEZ greatly increased BOEM’s environmental coverage. Faced with a very poorly studied and remote environment, BOEM supported a series of deep surveys (Carney, 2001) along the Atlantic and GoM continental margins in the 1980’s. These studies confirmed the existence of vast soft sediment habitats, but also recognized previously known live bottoms, and found unexpected chemosynthetic communities. Both deep live bottoms and chemosynthetic communities can be classed as sensitive habitats. It can be noted that the core participants in the propose work participated in these previous surveys in many ways.
The results of BOEM’s first study of upper slope chemosynthetic communities, Chemosynthetic Ecosystems Study (BOEM Report 95-2001) began the trend continuing to this day of submersible-based investigation in a mixed exploration and detailed-study mode. A second study, Stability and Change in Gulf of Mexico Chemosynthetic Communities (BOEM Report 2002-036) greatly increased knowledge of the ecology of these systems. BOEM-led investigations of coral communities in the GoM began with the Lophelia I study (BOEM Report ) which followed a similar pattern of exploration and experimentation. The ongoing Chemo III study (BOEM contract #) expanded this research into the deepest parts of the GoM and included the latest submersible technologies and techniques to greatly expand our ability to discover and characterize deep-water hard-ground habitats and communities. The scientific value of these studies was increased by the initiative of the core members who successfully sought competitive funds from NOAA, DOE, and NSF.
The work proposed herein is designed to meet BOEM’ information needs concerning the location and functioning of deep coral communities and the association with platforms and shipwrecks. Preliminary studies have shown that deep coral communities at the slope base are different from those on the upper slope in much the same way that the normal background fauna differ. Therefore, BOEM cannot simply extrapolate upper-slope data down the entire margin.
Geological Setting
The authigenic carbonates that are a part of nearly every fluid-gas expulsion site provide the hard substrate necessary for many deep marine sessile organisms, particularly corals. Consumption of hydrocarbons by microorganisms at expulsion sites is intimately associated with carbonate precipitation and the production of hard substrates. Aerobic methane oxidation produces CO2 and decreases pH, favoring dissolution of carbonates (Aloisi et al., 2002). Anaerobic microbial sulfate reduction using hydrocarbon substrates causes sulfate depletion and simultaneous bicarbonate and hydrogen sulfide enrichments in sediments. The increase in carbonate alkalinity of pore fluids produces calcium-magnesium carbonate by-products (Ritger et al, 1987; Roberts and Aharon, 1994). These carbonates take the form of distinct mounds with relief of up to 10 m, larger boulders and blocks, and low relief slabs and hardgrounds.
Recent manned submersible and ROV dives to the middle and lower continental slope confirm the existence of these hard substrates to the deepest parts of the slope. Direct observation and sampling of expulsion sites started in the mid-1980s on the upper slope.
We now know from analysis of 3D-seismic data and submersible-ROV dives that numerous expulsion sites with hard substrates provide habitat for deep water corals exist over the slope’s full depth range.
Deep Hardground Communities (Reefs, Rigs, and Wrecks)
In the Gulf of Mexico, deep-water corals are commonly found on seep-related authigenic carbonates, but have also been observed on anthropogenic structures, ship wrecks and oil platforms in particular. The most common species of reef-forming deep-water coral in the Gulf of Mexico (GoM) is Lophelia pertusa (=prolifera). This species was first recovered in the late 1800s by the U.S. Coast Survey Steamer Blake (Cairns 1978). L. pertusa “reefs” in the GoM were first reported from a deep water trawl taken by the M/V Oregon in 1955 (Moore and Bullis, 1960). Recently, submersible observations have located L. pertusa in numerous locations on the upper slope of the northern GoM (Schroeder 2002, Schroeder et al. 2005, Cordes et al. 2006, Cordes et al. 2008, CSA, 2007). The most extensive areas of L. pertusa reefs in the central GoM known to date are located on the upper De Soto Slope in BOEM lease block Viosca Knoll (VK) 826. There are more isolated colonies of L. pertusa in VK 862, in Mississippi Canyon (MC) 885 and on the upper Louisiana Slope in lease blocks Green Canyon (GC) 81, GC 184 (Bush Hill), GC 234, and GC 354 between 300 m and 540 m depth. In addition, L. pertusa was observed at the end of a dive in Garden Banks 535 at 580 m depth in 2003 (E.E.C. pers. obs.). Because the majority of investigations of hard substrata in the GoM have been directed towards chemosynthetic habitats, exploration focused on appropriate habitat for coral communities is very likely to expand this range both geographically and bathymetrically.
Deep-water corals have also been observed colonizing artificial substrates in the Gulf of Mexico. The most extensive currently known site of colonization is on the wreck of the Gulfpenn in MC 497. Two scleractinians, Lophelia pertusa and Pourtalosmilia conferta, were found on the Gulfpenn (Church et al. 2007). P. conferta was restricited to a solitary, medium size colony observed near the top of the starboard boom stanchion. L. pertusa was far more abundant, colonizing 12-15 percent of exposed surfaces and structures throughout the wreck. L. pertusa appears to have developed most successfully on surfaces or structures that have a vertical orientation (e.g. hull, bulwarks and sides of the superstructures) or that have an upright (e.g. davits, masts, booms and stacks), raised (e.g. catwalks and deck piping), or open (e.g. railings and rigging) construction. The most extensive coral growth is occurring along the railing, bulwarks and hull on the starboard side and on the deck piping and catwalks. At numerous locations clusters of adjacent colonies are coalescing in an initial phase of ‘thicket’ building. The largest development is a 6 to 7 m high by 3 to 3.5 m wide aggregate of at least 5 or 6 coalescing colonies. L. pertusa was also found living on the sediment adjacent to the hull and colonizing wreckage scattered about in the debris field northwest and west of the Gulfpenn.
Increasing industry activity in deepwater has resulted in the creation of numerous platforms in water depths exceeding 300 m. In areas where hard substrates are limiting, these platforms may significantly increase the potential range of corals and other hardground fauna. Growth of Lophelia pertusa has been noted on the Pompano platform in VK 989. In addition, the Joliet platform in GC 184 near Bush Hill and the Neptune platform near the large L. pertusa site in VK 826 are very likely to host coral populations. This study will focus on the exploration and characterization of these communities and examination of their potential connection to other coral populations and surrounding deep-water communities.
Deep-water Coral Biology and Ecology
L. pertusa develops from individual polyps, to larger “thickets”, to massive reef structures. It has separate sexes and exhibits seasonality in reproduction with spawning apparently occurring in September to October in the GoM (Brooke et al. 2005, Brooke and Schroeder 2007), and February in the Northeast Atlantic (Brooke et al. 2005, Waller and Tyler 2005). Gametes are released and fertilized externally, producing lecithotrophic planula larvae (Waller and Tyler 2005) which require hard substrata for settlement (Wilson 1979). Individual branches of coral grow slowly, and current estimates of linear extension rates range from 2-30 mm∙yr-1, although pioneer colonies on man-made substrates may grow much faster. (Mortensen and Rapp 1998, Gass and Roberts 2006). As the branches of the colony grow, new polyps are added via intra-tentacular budding (Cairns 1979). Small, young colonies consisting of living, white coral polyps are normally positioned along the edge of rocky outcrops or authigenic carbonates. As they grow, larger thickets of coral are usually comprised of a periphery of living coral surrounding a central dead portion of coral skeleton that serve as substrate for secondary settlement of L. pertusa and other species of deep-water corals (Wilson 1979). Larger, reef-like structures may cover extensive areas. The largest known continuous reef structure is roughly oval in shape covering 13 km along its axis and 300 m in diameter and consists of coral matrices up to 35 m thick (Freiwald et al. 1999). Carbon (14C) dating has placed the age of dead corals at the center of similar (but much smaller) structures in the GoM at over 40,000 years (Neumann et al. 1977).
Deep-water corals are important sources of habitat heterogeneity on continental shelves worldwide. They create habitat for a diverse group of associated fauna that may occur in abundances orders of magnitude above that on the surrounding seafloor (Jensen and Frederiksen 1992, Henry and Roberts 2007). In the Northeast Atlantic alone, over 1300 species have been recorded living in and on L. pertusa habitats in physical and photographic samples (Rogers 1999, Roberts et al. 2006, Henry and Roberts 2007). On two reef structures alone in Norway, 256 species were recorded from recovered coral blocks (Jensen and Frederiksen 1992). In the Porcupine Seabight in the Northeast Atlantic, 313 taxa were collected in 7 box core samples from coral mounds (Henry and Roberts 2007). The diversity of the community on these reefs rivals the diversity of many tropical zooxanthellate coral reefs (Rogers 1999). However, these studies all involved dredge and box core samples that include species that are likely not be intimately associated with coral structure but inhabit the sediments beneath the coral framework and in the case of dredges perhaps as much as 100s of meters away. In the GoM, 68 taxa of large macrofauna and megafauna (> 1 mm sieve size) were found in closely associated with L. pertusa in quantitative community collections obtained with custom collection devices deployed from a submersible (Cordes et al. 2008). Photographic surveys indicate that a variety of invertebrate and fish species occur primarily and possibly exclusively in these habitats (Ross and Quattrini 2007, CSA 2007, Lessard-Pilon, Cordes, and Fisher in prep). Some associated species appear to have very specific interactions with L. pertusa including the polychaete Eunice sp. which may help to assemble coral structure and the coralivorous gastropod Coralliophila sp. (Cordes et al. 2008; Becker, Cordes, and Fisher in prep).
While L. pertusa remains the best known deep-water corals species in the GoM in particular and the world in general, many other species of deep-water corals exist and are not as well studied. The number of described deep-water (defined as inhabiting aphotic waters greater than 200 m depth) scleractinian coral species now exceeds the number of described shallow water scleractinians (Cairns 2001). In the Gulf of Mexico, 63 species of azoozanthellate scleractinians have been reported (Cairns 1993). In addition to L. pertusa, 3 other species are known to form reef-like structures. Madrepora oculata is a cosmopolitan reef forming species which has been found down to 1500 m depth (Cairns 1978). Enallopsammia profunda is found to 2165m depth and is another common component of the deep-water reef-forming coral assemblage (Rogers 1999). Solenosmilia variabilis has been documented in waters to 3383 m (Cairns 1978). This species is a contributor to coral frameworks in the Atlantic (Rogers 1999) and is the most common deep water reef species on Southwest Pacific seamounts (Koslow et al. 2001). All three of these species were observed and collected at a recently discovered site in Green Canyon 852 during the ongoing BOEM/NOAA funded characterization of chemosynthetic communities in the deep Gulf of Mexico (Roberts et al. 2007). There are also at least 5 species of solitary scleractinians which contribute to the species richness of the deep-water coral community below 1000 m in the Gulf of Mexico (Cairns 1978).
In addition to scleractinian corals, a number of gorgonian and antipatharian corals are present in deep waters in the GoM (Cairns 1978, Cairns 1993). They can occur in the same areas with extensive development of scleractinian corals as well as in areas lacking reef-building hard corals. In either case these taxa add significant habitat heterogeneity and vertical relief. Gorgonians (Anthozoa: Octocorallia: Gorgonacea) are some of the most common and conspicuous sessile coral reef fauna in the shallow GoM and Caribbean Sea (Cairns 1977), and occur worldwide deeper than 4200 m (Bayer 1956). Gorgonians create habitat for associated species of fish (Heifetz 2002, Etnoyer & Warrenchuk 2007), invertebrates (Krieger & Wing 2002, Buhl-Mortensen and Mortensen 2005), and microbial fauna (Penn et al 2006). Gorgonians are also relatively long lived organisms, with single colonies of Primnoa reseadiformis estimated to be between 150 (Andrews et al. 2002) and 500 years old (Risk et al. 2002). Recent reviews of octocoral diversity in the GoM place the total number of species at 162 (Cairns and Bayer, in press). Gorgonacea in the Gulf occur from intermediate to abyssal depths (Isididae, 250-2800 m; Primnoidae 200-1400 m) (Etnoyer, unpublished data). The most common deep-water species include the endemic primnoid Callogorgia americana delta that forms extensive stands at many of the Green Canyon sites and on carbonate blocks in VK 826, and the broadly distributed bamboo coral Acanella eburnea. Precious corals (Coralliium spp.) are rare, but one colony was recently sampled at GC 852 during the Chemo III project.
Forty-two species of antipatharians have been recorded from the western Atlantic and about 30 of these have been found in the GoM (Cairns et al., 1993). The greatest species diversity occurs in a depth range of about 20 to 200 m, however, some GoM species have been reported from depths greater than 300 m, including Elatopathes abietina (Pourtales), Bathypathes patula Brook,Aphanipathes pedata Gray, Stylopathes columnaris (Duchassaing and Michelotti), Sibopathes macrospina Opresko, Tanacetipathes hirta (Gray), Parantipathes tetrasticha (Pourtales), andLeiopathes glaberrima (Esper). Other antipatharian taxa that might be expected to occur in deep waters in the GOM are species of the genera Chrysopathes spp., Heliopathes, and Acanthopathes. Although many antipatharian species form colonies of relatively small size, some are large enough to create habitat for many other marine organisms (see Yoklavich and Love, 2005). There is, however, very little information on the habitat forming species in the deep waters of the GoM, and very little is known about the distribution of antipatharians in the deep sea in general. In deep reef and upper slope habitats antipatharians can be a major component of the sessile benthic fauna, and it is likely that they play a similar role in the deep sea – particularly in locations characterized by distinct topographic features, hard bottom, and relatively fast currents, such as many of the hardgrounds of the GoM slope.
Deep-sea Environmental Issues and Impacts
The current lack of a significant deep-water fishery in the GoM also makes this an ideal setting for the study of deep-water coral habitats in relatively pristine condition. In other regions, the past and ongoing depletion of populations of deep water fishes with long life spans such as rockfish (Sebastesspp.), orange roughy (Hoplostethus atlanticus), and pelagic armourhead (Pseudopentaceros wheeleri) has resulted in significant damage to coral communities (Rogers 1999, Koslow et al. 2001, Roberts et al. 2002). The deepest commercial fisheries for bottom-dwelling fishes in the GoM remain in waters overlying the continental shelf. However, the presence of economically viable fishery species such as the longfin hake (Phycis chesteri), wreckfish (Polyprion americanus), blackbelly rosefish (Heliocolenus dactylopterus) and numerous grenadiers (Macrouridae) in deep waters (McEachran and Fechhelm 1998) and specifically at L. pertusa banks (Sulak et al. 2007) suggests that there may be current fishery activity that has been largely undocumented, and that a larger commercial deep water fishery in the GoM may yet develop.
The correlation between the distribution of deep-water corals and the location of deep water human activity has led to recent conservation efforts on the behalf of deep-water corals. For example, the Darwin Mounds off Scotland (Bett 2001, Masson et al. 2003) were recently proposed by the European Union as Special Areas of Conservation. The Lophelia reefs off Norway are protected under the Coral Protection Regulation of 1999 (Fossa et al. 2002). In the areas surrounding the Azores, Madeira, and Canary Islands, all bottom trawling is prohibited in depths > 200m. Off New Zealand and Australia, seamounts are closed to dredging and trawling, and a marine protected area (MPA) was established to protect the scleractinian Solensmilia variabilis (Koslow et al. 2001, Hall-Spencer et al. 2002). In 2005, the Aleutian Islands Habitat Conservation Area was established covering nearly 1 million km2 for deepwater coral protection off Alaska. Bottom trawling is restricted in two conservation areas in Atlantic Canada and in canyons near George’s Bank off the New England coast. Off the southeast U.S. coast, a MPA exists for Oculina varicosa off the coast of Florida and a large deep-water coral Habitat Area of Particular Concern has been proposed. In the GoM, the high-density L. pertusa portion of Viosca Knoll 826 has been proposed as a conservation area. To compliment these activities in the U.S., the 2007 reauthorization of the Magnuson-Stevens Fishery Conservation and Management Act called for the Deep Sea Coral Research and Technology Program to locate, map, and monitor activity in locations where deep-sea corals are known or likely to occur. The predictive capacity for determination of areas where corals are “likely to occur” will be developed as a prime focus of the proposed research here and will be a significant tool for future management of deep coral communities in the GoM.
Project Team
The Management Team
The Management Team includes a Program Manager, four Team Leaders, and a Data Manager. The qualifications of the designated individuals are well known. Data management is central to the success of all aspects of the program.
Key Personnel
|
Name |
Discipline |
Role |
Organization |
|
Dr. James M. Brooks |
Marine Geochemist |
Project Director |
TDI-Brooks Int’l Inc. |
|
Dr. Charles Fisher |
Biological Oceanography |
Biology Team Leader and Interdisciplinary Integration |
Penn State University |
|
Dr. Harry H. Roberts |
Marine Geology/Geophysics |
Geology/Geophysics/Physical Oceanography Team Leader |
Coastal Studies Institute, Louisiana State Univ. (LSU) |
|
Dr. Erik E. Cordes |
Marine Ecology |
Phylogeography/Taxonomy and Live Coral Studies Team Leader. |
TempleUniversity |
|
Dr. Chris German |
Open ocean geochemistry |
Sediment Trap deployment and analyses |
Woods Hole Oceanographic Institution |
|
Mr. Dan Warren |
Nautical Archaeology |
Archaeology-Shipwrecks |
C&C Technologies |
|
Dr. Ian R MacDonald |
Biological Oceanography |
Imaging/Fisheries Review |
Texas A&M University at Corpus Christi |
|
Dr. Bernie B. Bernard |
Marine Geochemist |
Lab Director/ Geochemical Studies |
TDI-Brooks Int’l Inc. |
|
Dr. Gary A. Wolff |
Biological Oceanographer |
Data Manager |
TDI-Brooks Int’l Inc. |
|
Ms. Liz Goehring |
Science communication and education |
Education Outreach |
Penn State University |
Associate Investigators/Co-PIs
| Name |
Discipline |
Role |
Organization |
|
Dr. Thomas J. McDonald |
Marine/Environmental Chemist |
Hydrocarbon Geochemistry |
TDI-Brooks Int’l Inc. |
| Dr. Susan Welsh |
Physical oceanographer |
Ocean Current Modeling |
Louisiana State Univ. (LSU) |
| Dr. Iliana Baums |
Coral Molecular Ecology |
Antipatharian Population Genetics and Aquarium Design |
Pennsylvania State Univ. |
|
Dr. Timothy Shank |
Molecular Ecology & Evolution |
Phylogeographic/Genetic Analysis of Coral-Associated Communities |
Woods Hole Oceanographic Institution |
| Mr. Rob Church |
Nautical Archaeology |
Archaeology-Shipwrecks |
C&C Technologies |
| Dr. Stephen Cairns |
Biology/Taxonomy |
Scleractinian Coral Taxonomy |
Smithsonian Institution |
|
Dr. William Schroeder |
Biology/Coral Ecology |
Corals – Shipwrecks/ Platforms |
DISL |
| Dr. Peter Etnoyer |
Biology/Taxonomy |
Octocoral Taxonomy |
Texas A&M University at Corpus Christi |
|
Dr. Dennis Opresko |
Biology/Taxonomy |
Antipatharian Taxonomy/Phylogeography |
Oak Ridge National Laboratories, Oak Ridge TN |
Taxonomic Collaborators
| Name |
Discipline |
Role |
Organization |
|
Dr. Sabine Stohr, |
Biology/Taxonomy |
Ophiuroid taxonomy |
Swedish Museum of Natural History |
|
Dr Daphne Fautine |
Biology/Taxonomy |
Anemone taxonomy |
University of Kansas |
|
Dr. Anders Waren, |
Biology/Taxonomy |
Gastropod taxonomy |
Swedish Museum of Natural History |
|
Stephen Hourdez |
Biology/Taxonomy |
Polychaete Taxonomy |
Statione Biologique de Roscoff, France |
|
Dr. Tom Shirley |
Biology/Taxonomy |
Crustacean taxonomy |
TAMU CC |
|
Maria Pia Miglietta |
Biology/Taxonomy |
Hydroid taxonomy |
Penn State University |
Support Staff
| Name |
Discipline |
Role |
Organization |
|
Ms. Suzanne Cardwell |
Administration |
TDI-Brooks’ Business Manager |
TDI-Brooks Int’l Inc. |
|
Mr. Michael Kullman |
Geographer |
GIS, navigation |
TDI-Brooks Int’l Inc. |
|
Mr. Cesar Arias |
Archaeology |
Geographer, Mosaics |
Texas A&M University |
|
Ms. Susan Wolff |
Editor/ Graphic artist |
Technical Editing/ Program Assistance |
TDI-Brooks Int’l Inc. |
|
Dr. Susanne McDonald |
Biological Oceanographer |
Laboratory QA and HSE |
TDI-Brooks Int’l Inc. |
Participants
|
Project Team |
Affiliation |
|
Phone |
|
Jim Brooks |
TDI-BI | Drjmbrooks@aol.com |
979.696-3634 |
|
Chuck Fisher |
Penn State | cfisher@psu.edu |
814.865-3365 |
|
Harry Roberts |
CSI-LSU | hrober3@lsu.edu |
225.578-2964 |
|
Dan Warren |
C&C Tech | djw@cctechnol.com | 832-461-4718 |
|
Rob Church |
C&C Tech | rob.church@cctechnol.com | 337- 735-3771 |
|
Ian MacDonald |
FSU | imacdonald@fsu.edu |
850-644-5498 |
|
Erik Cordes |
Temple | erik.cordes@temple.edu |
215-204-8876 |
|
Bernie Bernard |
TDI-BI | berniebernard@tdi-bi.com |
979.690-6287 |
| William Schroeder | UA | wschroeder@www.disl.org | 251-455-4010 |
|
Elizabeth Goehring |
Penn State |
exg15@psu.edu |
814- 863-6603 |
|
Gary Wolff |
TDI-BI | garywolff@tdi-bi.com |
979.693-3446 |
|
Chris German |
WHOI | cgerman@whoi.edu |
508 289 2853 |
|
Tim Shank |
WHOI | tshank@whoi.edu |
508 289 3761 |
|
Government |
|||
|
Greg Boland, COTR /CO |
MMS | gregory.boland@boem.gov |
703-787-1711 |
|
Emily McDonald |
NOAA/OE | Emily.McDonald@noaa.gov |
301-734-1003 |
Science Review Group
|
James Ray |
jray16@comcast.net |
|
Les Watling |
watling@hawaii.edu |
|
Gordon Watts |
iimr@coastalnet.com |
Cruises
Cruise 1
Cruise 2
Cruise 3
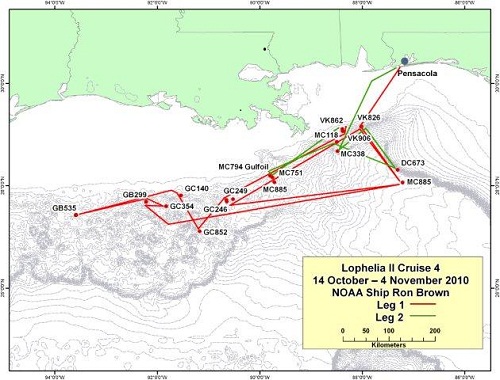
Cruise 4
Cruise 4
14 October – 4 November, 2010
![]()
Contact Us
TDI-Brooks International, Inc.
14391 S. Dowling Road
College Station, TX 77845
Email: info@tdi-bi.com
Phone: +1 (979) 693-3446



